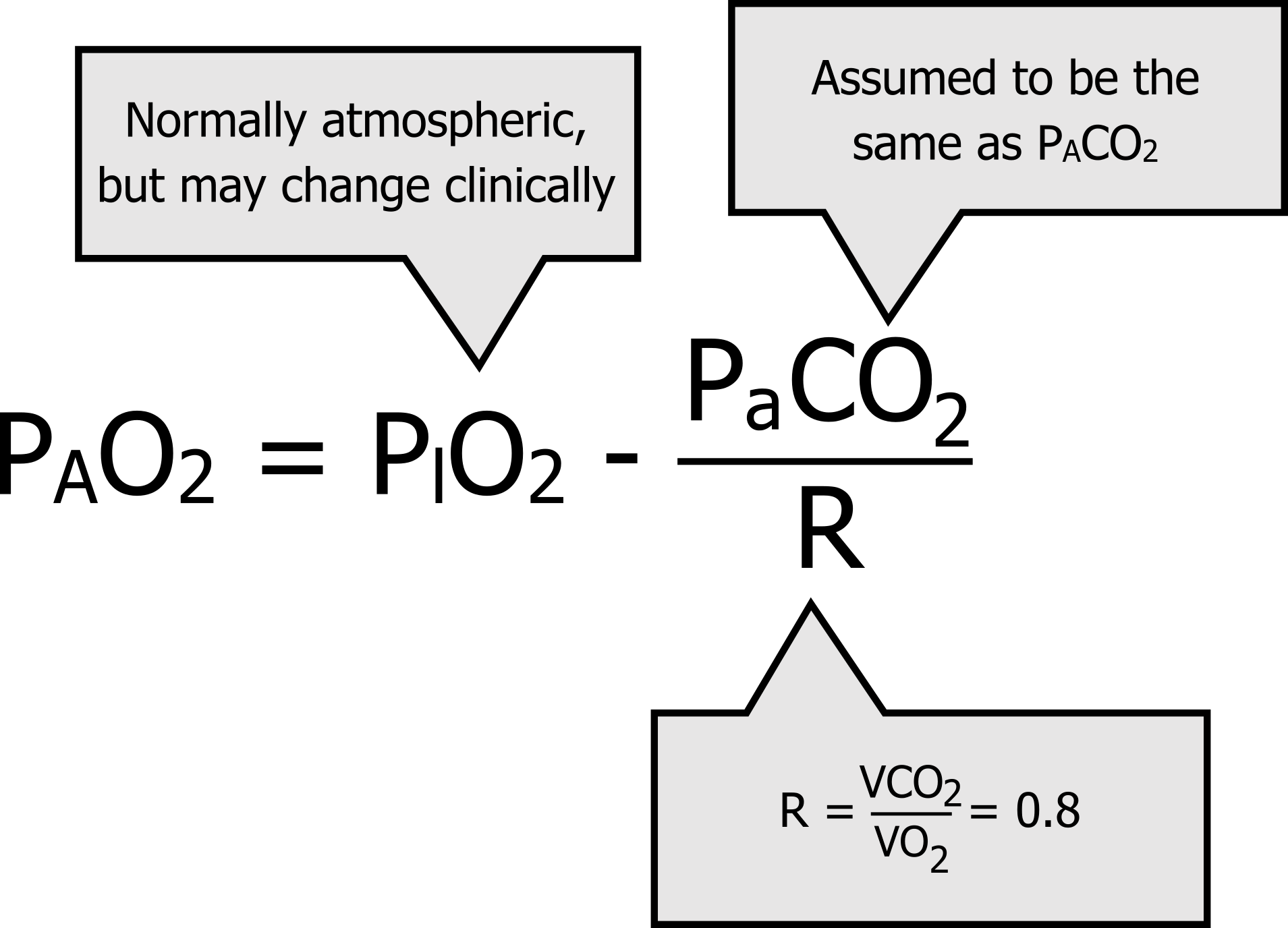
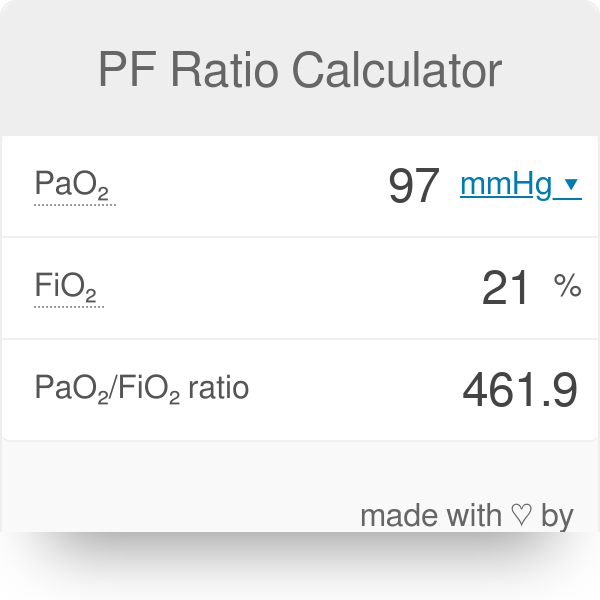
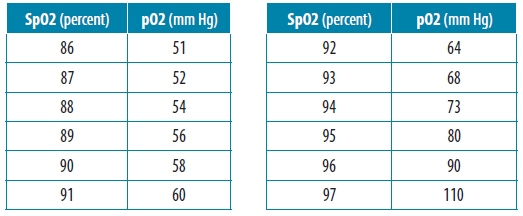
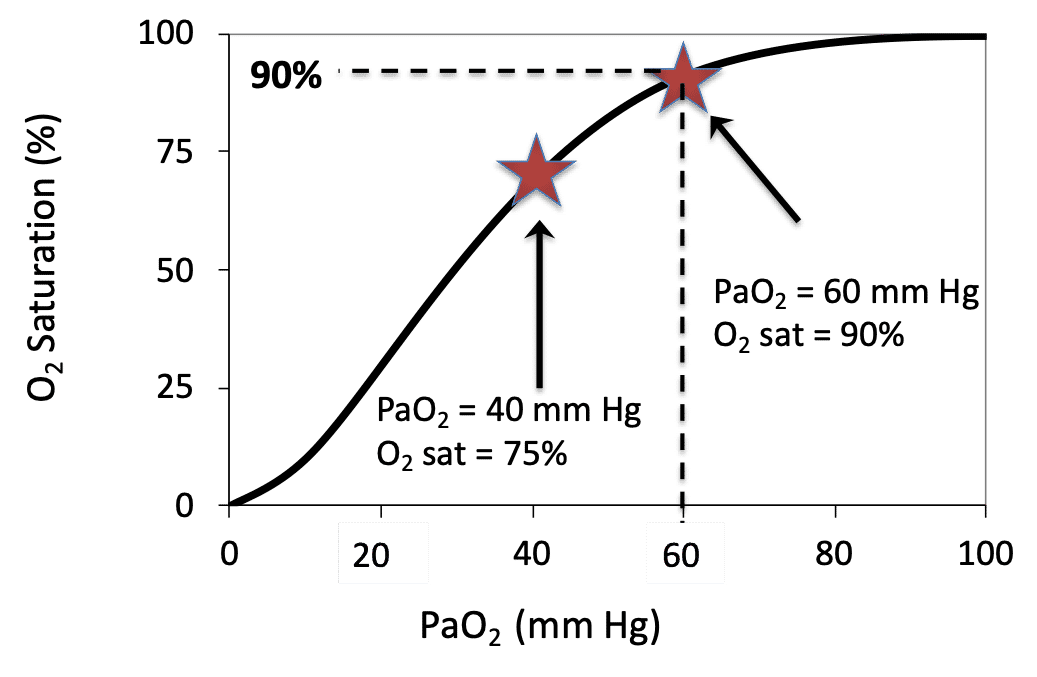
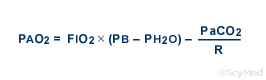SOLVED: A. What is the equation to calculate the partial pressure of a gas? B. Calculate the PaO2 for the air at 18,000 feet (atmospheric pressure 280 mm Hg). Show your equation/work.

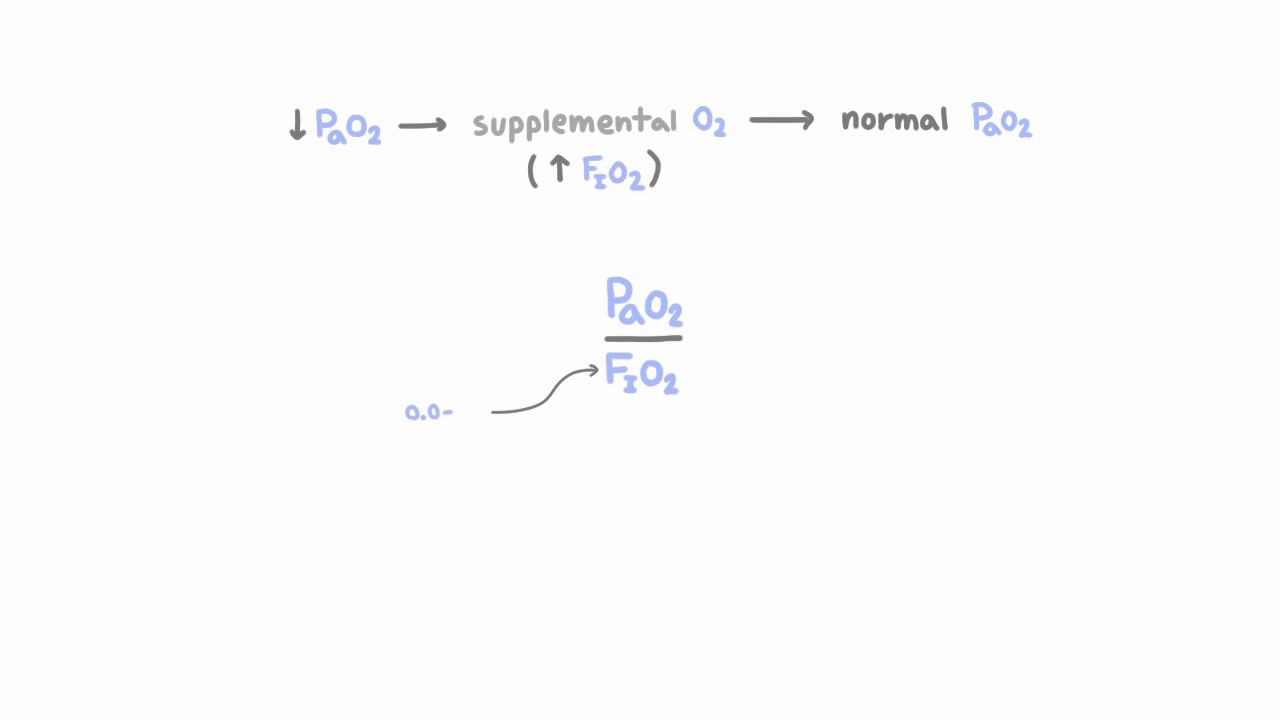
M Velia Antonini on Twitter: "Understanding gas analysis #eSMART2021 tutorial 🧵Starting from oxygenation: 🩸O2 content Hb bonded + dissolved in plasma 🩸lung capillary? assume Sat 100%; calculate PAO2 alveolar PO2 with alveolar